NICOTINIC RECEPTORS
Nicotinic receptors are a large family of cell membrane neurotransmitter receptors that are expressed in a variety of ways throughout the body. Diversity in structure and function makes nicotinic receptors important in multiple physiological pathways. This is largely why nicotinic receptors are implicated in multiple disorders including:
- Schizophrenia
- Parkinson’s Disease
- Epilepsy
- Alzheimer’s disease
- Addiction
- Autism
The fact that nicotinic receptors play a role in so many different disease states makes them a target for pharmacotherapy.
Nicotinic receptors are comprised of 5 subunit proteins that assemble inside the cell and migrate to the cell membrane. To date, nine alpha (α) subunits (2-10) and three beta (β) subunits (2-4) have been identified in nicotinic receptors in the central nervous system. Muscle type nicotinic receptors also contain delta and gamma subunits. The combinations of alpha and beta subunits that can be assembled in neuronal nicotinic receptors allow for marked diversity in structure and function. Consequently, there are many nicotinic receptor subtypes, which are individual types of nicotinic receptors comprised of unique combinations of alpha and beta subunits.
The focus of our research is centered on neuronal nicotinic receptors. When these receptors are stimulated by nicotine, they allow influx of calcium ions into the cell. This event triggers an action potential and the neurons release their stores of neurotransmitters.
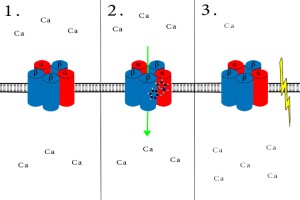
1. When a cell is at rest, calcium ions are evenly distributed across the membrane. 2. Following nicotine treatment, nicotinic receptors open their channel to allow calcium influx. 3. Influx of calcium ions initiates an action potential and subsequent neurotransmitter release.
Diversity in structure and function presents problems when trying to develop a drug for nicotinic receptors. Since drugs will affect different subtypes they will affect other functions that are not necessarily intended for therapy. Therefore, nicotinic receptor therapy is usually complicated with multiple side effects. This has prompted researchers and drug companies to identify compounds that will selectively target various nicotinic receptors in order to minimize side effects of treatment.
THE TARGET
Lung cancer is the number one preventable cause of death in the United States. Directly associated with smoking and nicotine addiction, lung cancer kills millions of people per year. Interestingly, a particular subtype of nicotinic receptor is implicated in the addictive properties of nicotine. This receptor is known as the α4β2 subtype. The reason that this particular subtype has been implicated in addiction is because it is expressed in the “addiction center” of the brain. The mesolimbic system containing the ventral tegmental area and nucleus accumbens is the center for dopamine signaling and is responsible for the body’s feeling of reward.
All addictive substances affect dopamine signaling in the brain because they cause the body to feel unnatural reward. Therefore, an addict will continue to seek out a substance regardless of negative consequences in order to experience its rewarding properties. These facts have directed our lab to developing a drug that is selective to the α4β2 nicotinic receptor in order to provide therapy for nicotine addicts while minimizing side effects.
DRUG DEVELOPMENT
The drug development process is a comprehensive examination of a substance’s ability to provide a therapeutic effect for a particular disease state. This process has been going on for centuries. For example, the Inca and Mayan civilizations would chew the leaves of the coca plant after discovering their analgesic properties. Today, the drug development process is highly regulated by the Food and Drug Administration (FDA), which oversees the production of all drugs and determines whether or not a drug is safe for the market.
The development process can take up to 10-12 years depending on the intended treatment and the source of the drug. Four phases make up the process starting with Phase 0 which is laboratory and animal testing of the drug candidate. Next is Phase 1, which is the beginning of human clinical trials and aims to determine whether the drug is toxic to humans. Phase 2 then examines whether the drug produces the intended effect on patients. Finally, Phase 3 validates the effectiveness of the drug on a large population of patients. Following Phase 3 the drug undergoes review by the FDA for approval for the market.
Our laboratory is in Phase 0 of the drug development process. We are examining a group of molecules that have been shown to have an affect on the α4β2 nicotinic receptor. Currently, we are using novel methods of drug screening and rational drug design to determine what compound will be the most effective treatment for the α4β2 receptor. In the future we intend to examine the effect of these drugs on animals and hopefully identify a compound that is a candidate for human trials.
Significance
Clinical Use
The primary significance of this research is rooted in the fact that the product will be a candidate for nicotine addiction therapy. No current smoking cessation aids have proven to be effective without the development of side effects. Recently, Chantix (Varenicline) has been popular in the industry because it aids in the cessation of smoking. However, Chantix is a non-selective partial agonist at α4β2 receptors and introduces many unwanted side effects by affecting multiple subtypes of nicotinic receptors. These side effects include, insomnia, constipation, mood and behavior changes, vivid and unusual dreams, and unpleasant taste in your mouth, among many others. Our research is significant because the mechanism by which our drugs work will allow for the cessation of smoking without the development of side effects.
Nicotinic Receptor Research
The development of selective modulators of nicotinic receptors is very significant in the scientific community. The physiological role of individual subtypes of nicotinic receptors is poorly understood and the development of selective modulators will provide insight into these physiological roles. To date, there have been very few selective nicotinic receptor ligands for any of the subtypes. The methodology behind our approach to developing a selective drug for the α4β2 receptor could be applied to any of the subtypes in order to develop selective modulators. We are not just developing a drug for nicotine addiction therapy, we are developing an approach to drug discovery at nicotinic receptors.

